Next: Solution of the 3D Up: Course Summary Previous: Solutions to the Radial Contents
The
Hydrogen (Coulomb potential) radial equation
is solved by finding the behavior at large
![]() ,
then finding the behavior at small
,
then finding the behavior at small
![]() ,
then using a power series solution to get
,
then using a power series solution to get
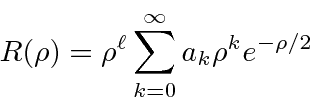
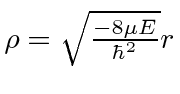 .
To keep the wavefunction normalizable the power series must terminate,
giving us our energy eigenvalue condition.
.
To keep the wavefunction normalizable the power series must terminate,
giving us our energy eigenvalue condition.
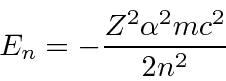
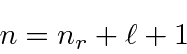
So a Hydrogen energy eigenstate
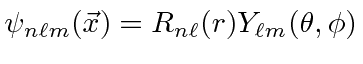 is described by three integer quantum numbers
with the requirements that
is described by three integer quantum numbers
with the requirements that
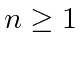 ,
,
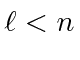 and also an integer, and
and also an integer, and
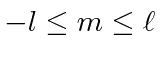 .
The ground state of Hydrogen is
.
The ground state of Hydrogen is
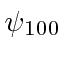 and has energy of -13.6 eV.
We compute several of the lowest energy eigenstates.
and has energy of -13.6 eV.
We compute several of the lowest energy eigenstates.
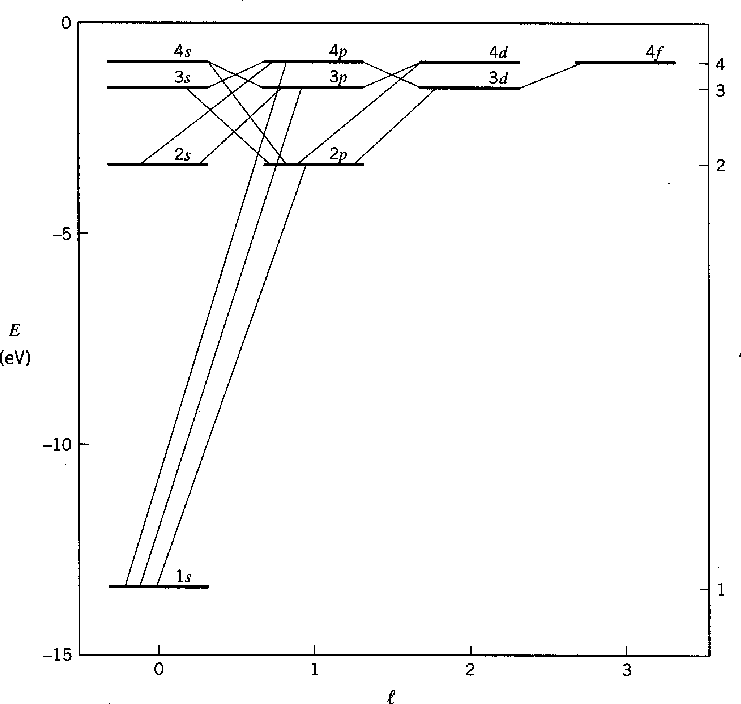
The diagram below shows the lowest energy bound states of Hydrogen and their typical decays.
Jim Branson 2013-04-22