Next: Piecewise Constant Potentials in Up: Course Summary Previous: Eigenfunctions, Eigenvalues and Vector Contents
As a concrete illustration of these ideas, we study the
particle in a box
(in one dimension).
This is just a particle (of mass
![]() )
which is free to move inside the walls of a box
)
which is free to move inside the walls of a box
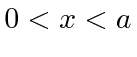 ,
but which cannot penetrate the walls.
We represent that by a potential which is zero inside the box and infinite outside.
We solve the Schrödinger equation inside the box and realize that the probability
for the particle to be outside the box, and hence the wavefunction there, must be zero.
Since there is no potential inside, the Schrödinger equation is
,
but which cannot penetrate the walls.
We represent that by a potential which is zero inside the box and infinite outside.
We solve the Schrödinger equation inside the box and realize that the probability
for the particle to be outside the box, and hence the wavefunction there, must be zero.
Since there is no potential inside, the Schrödinger equation is
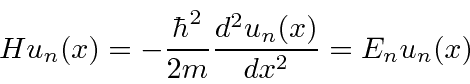
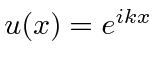 ,
,
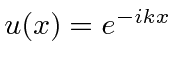 ,
,
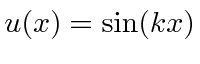 , and
, and
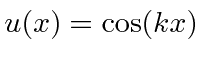 .
The wave function must be continuous though, so we require the boundary conditions
.
The wave function must be continuous though, so we require the boundary conditions
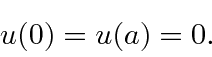
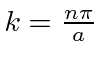 .
So the energy eigenfunctions are given by
.
So the energy eigenfunctions are given by
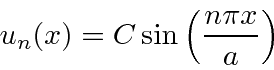
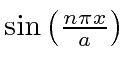 back into the Schrödinger equation,
we find that
back into the Schrödinger equation,
we find that
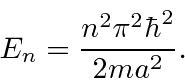
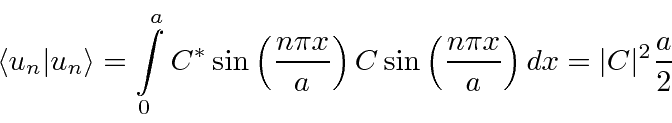
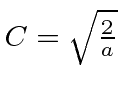 .
.
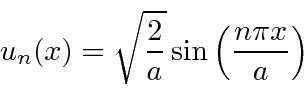
Jim Branson 2013-04-22