Next: The Rutherford Atom * Up: The Problems with Classical Previous: Black Body Radiation * Contents
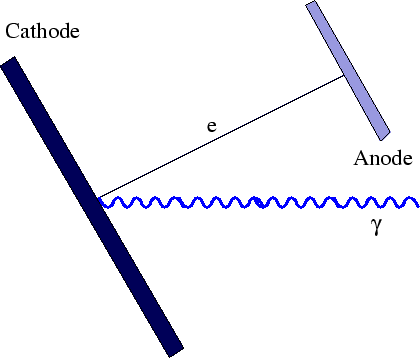
When light strikes a polished (metal) surface electrons are ejected.
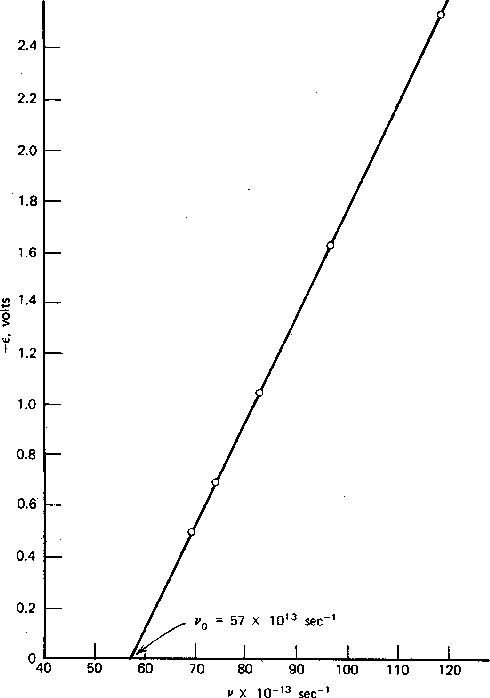
Measurements were made of the
maximum electron energy versus light frequency and light intensity.
Classical physics predicted that the electron energy should increase with intensity, as the electric field
increases.
This is not observed.
The electron energy is independent of intensity and depends linearly on the light frequency, as
seen the the figure above.
The kinetic energy of the electrons is given by Plank's constant times the light frequency minus a
work function
![]() which depends on the material.
which depends on the material.
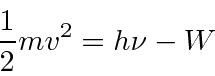
Jim Branson 2013-04-22