Next: The Stark Effect for Up: Examples Previous: H.O. with anharmonic perturbation Contents
We have solved the Hydrogen problem with the following Hamiltonian.
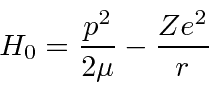
Now we want to find the correction to that solution if an
Electric field is applied to the atom.
We choose the axes so that the Electric field is in the z direction.
The perturbtion is then.
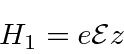
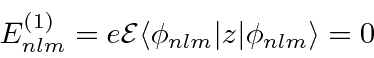
We therefore need to calculate the second order correction.
This involves a sum over all the other states.
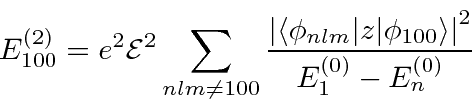
We need to compute all the matrix elements of z between the ground state and
the other Hydrogen states.
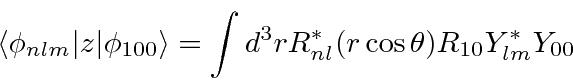
We can do the angular integral by converting the
![]() term into a spherical
harmonic.
term into a spherical
harmonic.
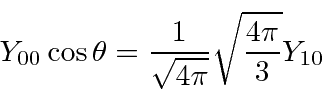
The we can just use the orthonormality of the spherical harmonics to do the angular integral, leaving us with a radial integral to do.
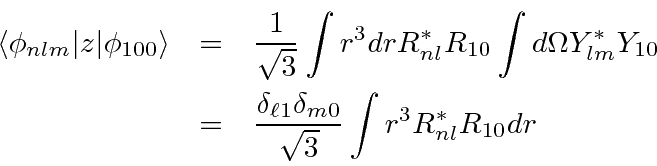
The radial part of the integral can be done with some work, yielding.
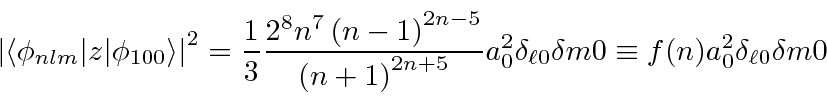
We put this back into the sum.
The Kronecker deltas eliminate the sums over
![]() and
and
![]() .
We write the energy denominators in terms of the Bohr radius.
.
We write the energy denominators in terms of the Bohr radius.
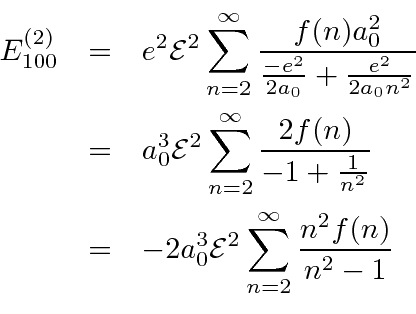
This is all a little dissatisfying because we had to insert the general formula for
the radial integral and it just goes into a nasty sum.
In fact, we could just start with the first few states to get a good idea of
the size of the effect.
The result comes out to be.
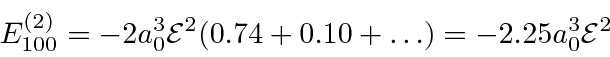
Now we compute
![]() , the electric dipole moment of the atom which
is induced by the electric field.
, the electric dipole moment of the atom which
is induced by the electric field.
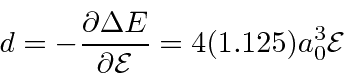
Jim Branson 2013-04-22