Next: Homework 9 Up: Homework Problems 130A Previous: Homework 7 Contents
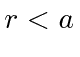 and
and 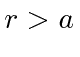 .
What are the limits for
.
What are the limits for
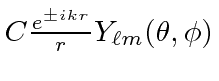 .
.
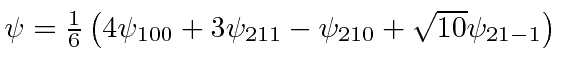 .
What are the possible energies that can be measured and what are the probabilities of each?
What is the expectation value of
.
What are the possible energies that can be measured and what are the probabilities of each?
What is the expectation value of
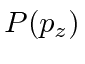 , the probability distribution of
, the probability distribution of 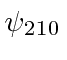 ?
You may find the expansion of
?
You may find the expansion of
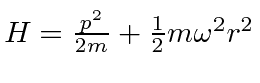 has been solved in the notes, using the same techniques as we used for Hydrogen.
Use the recursion relations derived there to write out the wave functions
has been solved in the notes, using the same techniques as we used for Hydrogen.
Use the recursion relations derived there to write out the wave functions
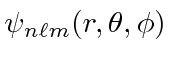 for the three lowest energies.
You may write them in terms of the standard
for the three lowest energies.
You may write them in terms of the standard 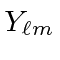 but please write out the radial parts of the wavefunction completely.
Note that there is a good deal of degeneracy in this problem so the three lowest energies actually means 4 radial wavefunctions
and 10 total states.
Try to write the solutions
but please write out the radial parts of the wavefunction completely.
Note that there is a good deal of degeneracy in this problem so the three lowest energies actually means 4 radial wavefunctions
and 10 total states.
Try to write the solutions 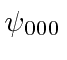 and
and 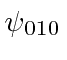 in terms of the solutions in cartesian coordinates with the same energy
in terms of the solutions in cartesian coordinates with the same energy
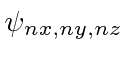 .
.
Jim Branson 2013-04-22