Next: The QED LaGrangian and Up: Dirac Equation Previous: The Quantized Dirac Field Contents
The term Vacuum Polarization is descriptive of the effect.
A charged particle will polarize the vacuum in a way analogous to the way a dielectric is polarized.
A virtual electron positron pair in the vacuum will be affected by the charge.
If the original charged source is a nucleus for example, the virtual electron will be attracted and the virtual positron repelled,
causing a net polarization of the vacuum which screens the nuclear charge.
At very short distances from the nucleus, the bare charge is seen,
while at long distances the screening is important.
This causes the basic coupling
![]() to vary a bit with distance and therefore with energy.
This polarization of the vacuum is similar to the polarization of a dielectric material.
In this case, what is being polarized are the virtual electrons and positrons in the vacuum.
Of course other particles than the electron can be polarized in the vacuum so the
energy variation of the coupling ``constant'' is an interesting subject for research.
to vary a bit with distance and therefore with energy.
This polarization of the vacuum is similar to the polarization of a dielectric material.
In this case, what is being polarized are the virtual electrons and positrons in the vacuum.
Of course other particles than the electron can be polarized in the vacuum so the
energy variation of the coupling ``constant'' is an interesting subject for research.
The effect of vacuum polarization on Hydrogen would be to lower the energy of
![]() states relative to others
since they are close to the nucleus and therefore see an unscreened charge.
This effect is actually rather small even compared to the Lamb shift and of opposite sign.
Vacuum Polarization has larger effects at higher energies at which shorter distances are probed.
In fact we can say that the electromagnetic coupling varies slowly with the energy scale,
increasing (logarithmically) at higher energies.
This is referred to as the running of the coupling constant.
states relative to others
since they are close to the nucleus and therefore see an unscreened charge.
This effect is actually rather small even compared to the Lamb shift and of opposite sign.
Vacuum Polarization has larger effects at higher energies at which shorter distances are probed.
In fact we can say that the electromagnetic coupling varies slowly with the energy scale,
increasing (logarithmically) at higher energies.
This is referred to as the running of the coupling constant.
We can get some qualitative understanding of the origin of Zitterbewegung from the idea of virtual pair production in the field of the nucleus. The diagram below shows a photon from the Coulomb field of the nucleus producing an electron positron pair. The original real electron from the atom then anihillates with the positron, coupling to another field photon. The electron from the pair is left over and becomes the new atomic electron, however, it need not be in the same place as the original electron.
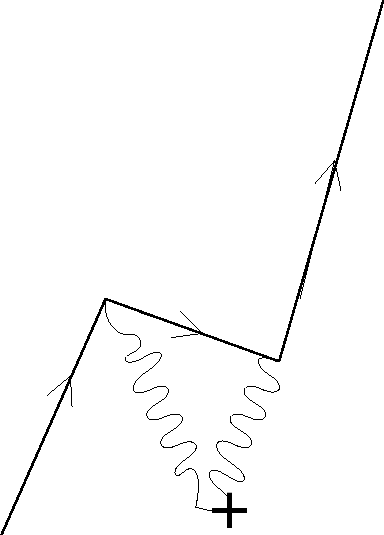
We can estimate the distance an electron might jump as it undergoes this process.
First the time for which the virtual pair exists can be estimated from the uncertainty principle.
Energy conservation is violated by
![]() at least so
at least so
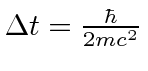 (which is approximately the reciprocal of the Zitterbewegung frequency).
The distance the electron appears to jump then is of the order of
(which is approximately the reciprocal of the Zitterbewegung frequency).
The distance the electron appears to jump then is of the order of
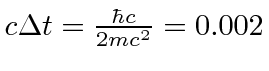 Angstroms.
This is the aproximate size of the fast back and forth motion of Zitterbewegung.
Angstroms.
This is the aproximate size of the fast back and forth motion of Zitterbewegung.
Jim Branson 2013-04-22