Next: LASERs Up: Phenomena of Radiation Theory Previous: Phenomena of Radiation Theory Contents
Mössbauer discovered that atoms in a crystal need not recoil significantly.
In fact, the whole crystal, or at least a large part of it may recoil,
making the energy shift very small.
Basically, the atom emitting an x-ray is in a harmonic oscillator (ground) state bound to the rest of the crystal.
When the x-ray is emitted, there is a good chance the HO remains in the ground state.
An analysis shows that the probability is approximately
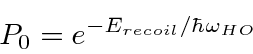
The Mössbauer effect has be used to measure the gravitational red shift on earth.
The red shift was compensated by moving a detector, made from the same material as the emitter,
at a velocity (should be equal to the free fall velocity).
The blue shift was measured to be
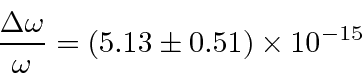
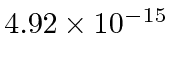 was expected based upon the general principle of equivalence.
was expected based upon the general principle of equivalence.
Jim Branson 2013-04-22