Next: Homework Problems Up: Examples Previous: Nitrogen Ground State Contents
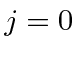 as a base.
It has four valence 2P electrons which we will treat as two valence 2P holes.
Hund's first rule , maximum total
as a base.
It has four valence 2P electrons which we will treat as two valence 2P holes.
Hund's first rule , maximum total
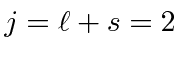 .
So the ground state is
.
So the ground state is
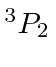 .
.
|
|
e |
| 1 |
|
| 0 |
|
| -1 |
|
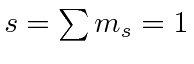 |
|
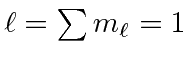 |
|
Jim Branson 2013-04-22