We will now add one parameter to the hydrogenic ground state wave function
and optimize that parameter to minimize the energy.
We could add more parameters but let's keep it simple.
We will start with the hydrogen wavefunctions but allow for the fact that one electron
``screens'' the nuclear charge from the other.
We will assume that the wave function changes simply by the replacement
Of course the
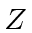 in the Hamiltonian doesn't change.
in the Hamiltonian doesn't change.
So our ground state trial function is
Minimize the energy.
We can recycle our previous work to do these integrals.
First, replace the
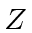 in
in
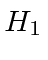 with a
with a
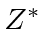 and put in a correction term.
This makes the
and put in a correction term.
This makes the
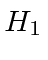 part just a hydrogen energy.
The correction term is just a constant over
part just a hydrogen energy.
The correction term is just a constant over
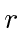 so we can also write that in terms of the
hydrogen ground state energy.
so we can also write that in terms of the
hydrogen ground state energy.
Then we reuse the perturbation theory calculation to get the
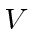 term.
term.
Use the variational principle to determine the best
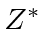 .
.
Putting these together we get our estimate of the ground state energy.
Now we are within a few percent.
We could use more parameters for better results.
Jim Branson
2013-04-22
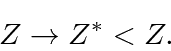
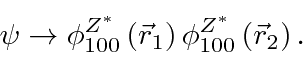
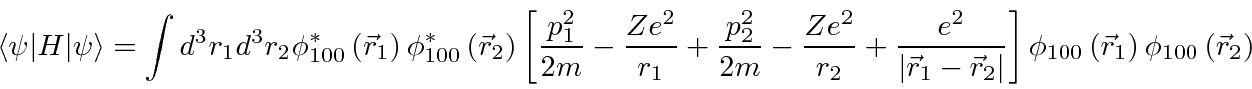
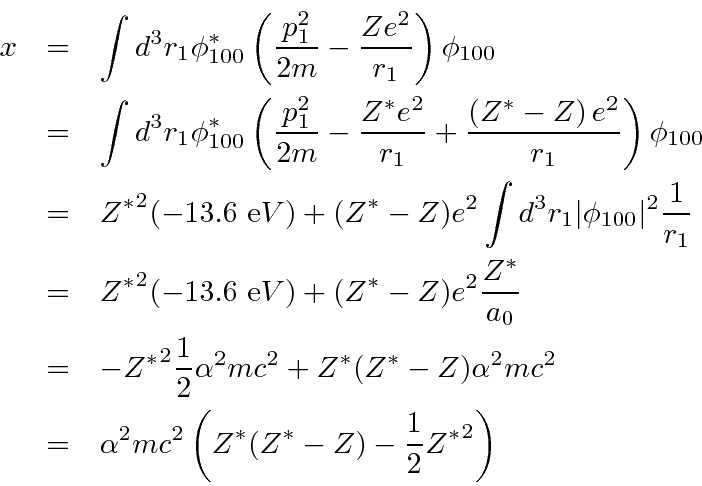
![]() term.
term.
![\begin{eqnarray*}
\left<\psi \vert H\vert\psi\right> &=& 2[x]+{5\over 4}Z^*\left...
...r 2}\alpha^2 mc^2\left[-2{Z^*}^2 + 4ZZ^* - {5\over 4}Z^* \right]
\end{eqnarray*}](img3308.png)
![]() .
.
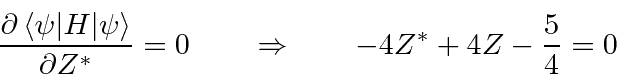
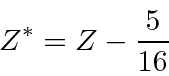
![\begin{eqnarray*}
\left<\psi \vert H\vert\psi\right> &=& -{1\over 2}\alpha^2 mc^...
...\right)^2\right] = -77.38 {\mathrm eV} \\
(really -78.975 eV).
\end{eqnarray*}](img3311.png)