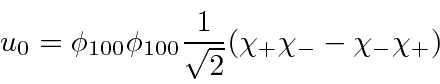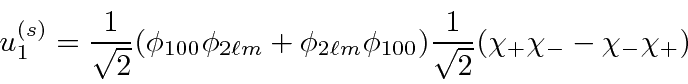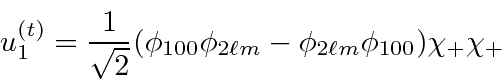We can use the hydrogenic states to begin to understand Helium.
The Hamiltonian has the same terms as Hydrogen but has a large
perturbation due to the repulsion between the two electrons.
We can write this in terms of the (
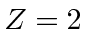 ) Hydrogen Hamiltonian for each electron plus a
perturbation,
) Hydrogen Hamiltonian for each electron plus a
perturbation,
where
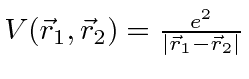 .
Note that
.
Note that
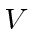 is about the same size as the the rest of the Hamiltonian so
first order perturbation theory is unlikely to be accurate.
is about the same size as the the rest of the Hamiltonian so
first order perturbation theory is unlikely to be accurate.
For our zeroth order energy eigenstates, we will use
product states of Hydrogen wavefunctions.
These are not eigenfunctions of H because of
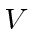 , the electron coulomb repulsion term.
Ignoring
, the electron coulomb repulsion term.
Ignoring
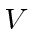 , the problem separates into the energy for electron 1 and the energy for
electron 2 and we can solve the problem exactly.
, the problem separates into the energy for electron 1 and the energy for
electron 2 and we can solve the problem exactly.
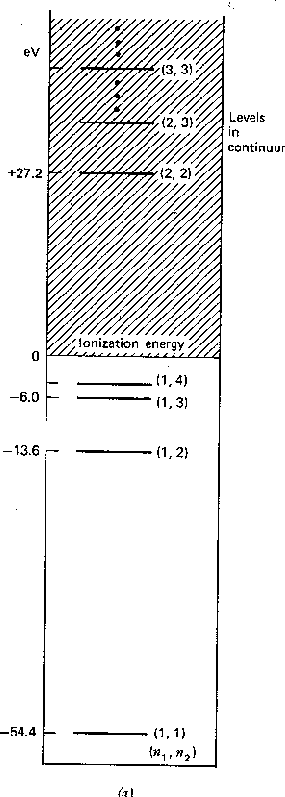
We can write these zeroth order energies in terms of the principal
quantum numbers of the two electrons,
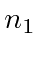 and
and
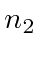 .
Recalling that there is a factor of
.
Recalling that there is a factor of
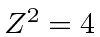 in these energies compared to hydrogen, we get
in these energies compared to hydrogen, we get
Note that
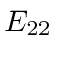 is above ionization energy,
so the state can decay rapidly by ejecting an electron.
is above ionization energy,
so the state can decay rapidly by ejecting an electron.
Now let's look at the (anti) symmetry of the states of two identical electrons.
For the ground state, the spatial state is symmetric,
so the spin state must be antisymmetric
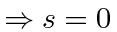 .
.
For excited states, we can make either symmetric or antisymmetric space states.
The first state is
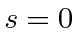 or spin singlet.
The second state is
or spin singlet.
The second state is
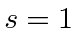 or spin triplet and has three
or spin triplet and has three
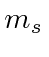 states.
Only the +1 state is shown.
Because the large correction due to electron repulsion is much larger for symmetric
space states, the spin of the state determines the energy.
states.
Only the +1 state is shown.
Because the large correction due to electron repulsion is much larger for symmetric
space states, the spin of the state determines the energy.
We label the states according to the spin quantum numbers, singlet or triplet.
We will treat V as a perturbation.
It is very large, so first order perturbation theory will be quite inaccurate.
Jim Branson
2013-04-22
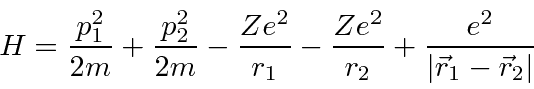
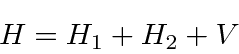
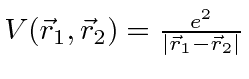 .
Note that
.
Note that
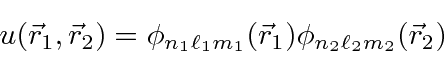
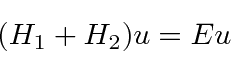
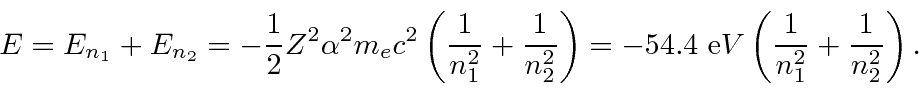
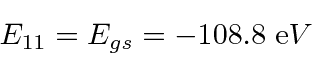
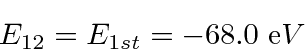
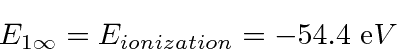
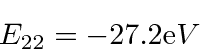
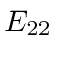 is above ionization energy,
so the state can decay rapidly by ejecting an electron.
is above ionization energy,
so the state can decay rapidly by ejecting an electron.
![]() .
.