Next: Importance of Unpaired Valence Up: Molecular Physics Previous: The Ion Contents
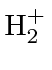 Hamiltonian,
the repulsion between electrons,
plus a correction term for double counting the repulsion between protons.
Hamiltonian,
the repulsion between electrons,
plus a correction term for double counting the repulsion between protons.
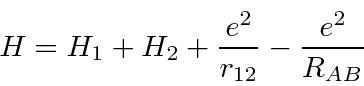
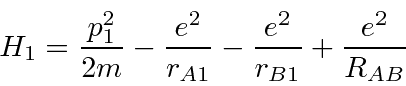
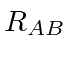 and the energy.
and the energy.
We will again use symmetric electron wavefunctions,
![\begin{displaymath}\bgroup\color{black}\psi(r_1,r_2)
= {1\over {2[1+S(R_{AB})]}...
...t[ \psi_A(\vec{r_2}) + \psi_B(\vec{r_2})\right] \chi_{s}\egroup\end{displaymath}](img3484.png)
The space symmetric state will be the ground state as before.
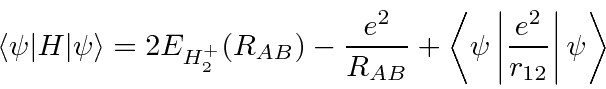
| Distance | Energy | |
| Calculated | 0.85 Å | -2.68 eV |
| Actual | 0.74 Å | -4.75 eV. |
wIth a multiterm wavefunction, we could get good agreement.
Jim Branson 2013-04-22